Editorial Policies
Focus and Scope
Statement
SciMedicine Journal is a multidisciplinary, an open-access, internationally double-blind peer-reviewed journal with a focus on practical applications of theory, research and experience and provides a bridge between academic knowledge, illness experience, wellbeing and health care practice.
Special Issues deal with more focused topics with high current interest falling within the scope of the journal in which they are published. Special Issue proposals are welcome at any time during the year.
Publishing Schedule
SMJ has been publishing four issues per year (Quarterly) since 2019. The exact schedule of publication for each issue is as the following:
1. March
2. June
3. September
4. December
Aims & Scope
SciMedicine Journal aims to publish the best research studies on international medicine sciences in a form that is accessible to practitioners and policy-makers as well as to an academic audience. Alongside its main focus, the journal aims a special focus on health in developing countries, which highlight the common problems of the region and their possible solutions. The SciMedicine Journal (SMJ) will consider multidisciplinary and emerging research submissions in the following areas:
|
|
|
|
|
|
|
|
|
|
|
|
Section Policies
Research Articles
 Open Submissions
Open Submissions Indexed
Indexed Peer Reviewed
Peer Reviewed
Review Articles
 Open Submissions
Open Submissions Indexed
Indexed Peer Reviewed
Peer Reviewed
Case Report
 Open Submissions
Open Submissions Indexed
Indexed Peer Reviewed
Peer Reviewed
Special Issue "COVID-19"
 Open Submissions
Open Submissions Indexed
Indexed Peer Reviewed
Peer Reviewed
Commentary
 Open Submissions
Open Submissions Indexed
Indexed Peer Reviewed
Peer Reviewed
Letter to the Editor
 Open Submissions
Open Submissions Indexed
Indexed Peer Reviewed
Peer Reviewed
Others
 Open Submissions
Open Submissions Indexed
Indexed Peer Reviewed
Peer Reviewed
Open Access Policy
This journal provides immediate open access to its content on the principle that making research freely available to the public supports a greater global exchange of knowledge.
Everyone has a right to "download, reuse, re-print, modify, distribute and/or copy" the published papers.
Archiving
This journal utilizes the LOCKSS system to create a distributed archiving system among participating libraries and permits those libraries to create permanent archives of the journal for purposes of preservation and restoration.
Abstracting, Indexing and Membership
Allegations of Misconduct
Plagiarism
Plagiarism includes, but is not limited to:
- Directly copying text from other sources without attribution
- Copying ideas, images, or data from other sources without attribution
- Reusing text from your own previous publications without attribution or agreement of the editor
- Exception: Reusing text from the Methods section in the author’s previous publications, with attribution to the source, is acceptable.
Using an idea from another source with slightly modified language without attribution.
If plagiarism is detected during the peer review process, the manuscript may be rejected. If plagiarism is detected after publication, we may issue a correction or retract the paper, as appropriate.
All manuscripts under review or published with the SciMedicine Journal are subject to screening using "iThenticate" software
Data fabrication
This concerns the making up of research findings
- Suspected fabricated data in a submitted manuscript: (https://publicationethics.org/files/Fabricated%20data%20A.pdf)
- Suspected fabricated data in a published manuscript: (https://publicationethics.org/files/Fabricated%20data%20B.pdf).
Data falsification
Manipulating research data with the intention of giving a false impression. This includes manipulating images (e.g. micrographs, gels, radiological images), removing outliers or “inconvenient” results, changing, adding or omitting data points, etc.
Duplicate submissions:
Duplicate submission is a situation whereby an author submits the same or similar manuscripts to two different journals at the same time either within Academic Journals or any other publisher. This includes the submission of manuscripts derived from the same data in such a manner that there are no substantial differences in the manuscripts. Duplicate submission also includes the submission of the same/similar manuscript in different languages to different journals.
Publication Ethics and Malpractice Statement
The ethical policy of SciMedicine Journal is based on the Committee on Publication Ethics (COPE) guidelines and complies with International Committee of SMJ Editorial Board codes of conduct. Readers, authors, reviewers and editors should follow these ethical policies once working with SMJ. The ethical policy of SMJ is liable to determine which of the typical research papers or articles submitted to the journal should be published in the concerned issue. For information on this matter in publishing and ethical guidelines please visit http://publicationethics.org
Principles of Transparency and Publication Ethics
Peer review process: SMJ is a double-blind peer-reviewed electronic quarterly publication concerned with all aspects of Medical Sciences. This process, as well as any policies related to the journal’s peer review procedures, is clearly described on the journal’s Web site (https://www.scimedjournal.org/index.php/SMJ/about/editorialPolicies#custom-5).
Governing Body: SMJ has a very strong editorial board, whose members are recognized experts in the subject areas included within the journal’s scope. The full names and affiliations of the journal’s editors are provided on the journal’s Web site (https://www.scimedjournal.org/index.php/SMJ/about/editorialTeam).
Identification of and dealing with allegations of research misconduct: Editor-in-Chief takes reasonable steps to identify and prevent the publication of papers where research misconduct has occurred, including plagiarism, citation manipulation, and data falsification/fabrication, among others.
Website: A journal’s Website (https://www.scimedjournal.org/) contains that care has been taken to ensure high ethical and professional standards.
Contact information: Journal is provided the contact information for the editorial office of SMJ (https://www.scimedjournal.org/index.php/SMJ/about/contact).
Name of journal: The Journal name of SciMedicine Journal (SMJ) has unique and not be one that is easily confused with another journal.
Conflicts of interest: Authors are requested to evident whether impending conflicts do or do not exist while submitting their articles to SMJ through Conflict of Interest Disclosure form (https://www.scimedjournal.org/Guideline/Conflict%20of%20Interest.docx).
Declaration of Competing Interests: The editor must not be involved in decisions about papers, which she/he has written him/herself or have been written by family members or colleagues or which relate to products or services in which the editor has an interest. Further, any such submission must be subject to all of the journal’s usual procedures, peer review must be handled independently of the relevant author/editor and their research groups, and there must be a clear statement to this effect on any such paper that is published.
Acknowledgments: All contributors who do not meet the criteria for authorship should be listed in an ‘Acknowledgements’ section. Examples of those who might be acknowledged include a person who provided purely technical help or writing assistance, or a department chair who provided only general support.
Disclosure of Funding Sources: SMJ authors are required to declare what support they received to carry out their research. Declaring funding sources acknowledges funders’ contributions, fulfills funding requirements and promote greater transparency in the research process.
* Note: SMJ will not consider for publication manuscripts in which any of the research costs or authors' salaries have been funded, in whole or in part, by a tobacco company.
Duties and Responsibilities of Editors
(http://publicationethics.org/files/Code_of_conduct_for_journal_editors_Mar11.pdf)
- The Editors of the journal should have the full authority to reject/accept a manuscript.
- The Editors of the journal should maintain the confidentiality of submitted manuscripts under review or until they are published.
- The Editor-in-Chief should make a decision on submitted manuscripts, whether to be published or not with other editors and reviewers
- The Editors of the journal should preserve the anonymity of reviewers.
- The Editors of the journal should disclose and try to avoid any conflict of interest.
- The Editors of the journal should maintain academic integrity and strive to meet the needs of readers and authors.
- The Editors of the journal should be willing to investigate plagiarism and fraudulent data issues and willing to publish corrections, clarifications, retractions, and apologies when needed.
- The Editors of the journal should have the limit themselves only to the intellectual content.
- The Editors of the journal must not disclose any information about submitted manuscripts to anyone other than the corresponding author, reviewers, potential reviewers, other editorial advisers, and the publisher, as appropriate.
- Unpublished materials disclosed in a submitted paper will not be used by the editor or the members of the editorial board for their own research purposes without the author's explicit written consent.
- Guest editors play a vital role in acquiring content and leading the review process for special issue publications.
Duties and Responsibilities of Reviewers
(http://publicationethics.org/files/u7140/Peer%20review%20guidelines.pdf)
- The Reviewers of the journal should assist the Editors in taking the decision for publishing the submitted manuscripts.
- The Reviewers should maintain the confidentiality of manuscripts, which they are invited to review.
- The Reviewers should provide comments in time that will help editors to make a decision on the submitted manuscript to be published or not.
- The Reviewers are bound to treat the manuscript received for peer reviewing as confidential, and must not use the information obtained through peer review for personal advantage.
- The Reviewers comments against each invited manuscript should be technical, professional and objective.
- The Reviewers should not review the manuscripts in which they have found conflicts of interest with any of the authors, companies, or institutions.
- The Reviewers should disclose and try to avoid any conflict of interest.
Authors of SciMedicine Journal must confirm the following:
- Submitted manuscripts must be the original work of the author(s),
- The submitting corresponding author is responsible for ensuring that the manuscript article's publication has been approved by all the other coauthors.
- Only unpublished manuscripts should be submitted,
- All authors have agreed to allow the corresponding author to serve as the correspondent with the editorial office, to review the edited manuscript and proof,
- Acknowledge the sources of data used in the development of the manuscript,
- All listed authors must have made a significant scientific contribution to the research in the manuscript and approved all its claims,
- All errors discovered in the manuscript after submission must be swiftly communicated to the Editor,
- All authors must know that that the submitted manuscripts under review or published with SMJ are subject to screening using Plagiarism Prevention Software. Plagiarism is a serious violation of publication ethics.
The SciMedicine Journal follows The International Committee of Medical Journal Editors (ICMJE) recommendations for the Conduct, Reporting, Editing, and Publication of Scholarly Work in Medical Journals (ICMJE Recommendations 2019) with regards to authorship, contributorship and group authorship.
Ethical Guidelines
Research Involving Human Subjects
When reporting studies that involve human participants, authors should include a statement that the studies have been approved by the appropriate institutional and/or national research ethics committee and have been performed in accordance with the ethical standards as laid down in the 1975 Declaration of Helsinki (https://www.wma.net/what-we-do/medical-ethics/declaration-of-helsinki/), revised in 2013, and its later amendments or comparable ethical standards.
If doubt exists whether the research was conducted in accordance with the 1975 Helsinki Declaration or comparable standards, the authors must explain the reasons for their approach, and demonstrate that the independent ethics committee or institutional review board explicitly approved the doubtful aspects of the study. At a minimum, a statement including the project identification code, date of approval, and name of the ethics committee or institutional review board should be stated in Section ‘Ethical Approval’ of the article.
Example of an ethical statement: "All subjects gave their informed consent for inclusion before they participated in the study. The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of XXX (Project identification code)."
Use of Animals in Research
The welfare of animals used for research must be respected. When reporting experiments on animals, authors should indicate whether the international, national, and/or institutional guidelines for the care and use of animals have been followed, and that the studies have been approved by a research ethics committee at the institution or practice at which the studies were conducted (where such a committee exists).
SciMedicine Journal endorse the ARRIVE guidelines (https://arriveguidelines.org/arrive-guidelines) for reporting experiments using live animals. Authors and reviewers can use the ARRIVE guidelines as a checklist, which can be found at: https://arriveguidelines.org/resources/author-checklists.
Research Involving Cell Lines
Methods sections for submissions reporting on research with cell lines should state the origin of any cell lines. For established cell lines, the provenance should be stated and references must also be given to either a published paper or to a commercial source. If previously unpublished de novo cell lines were used, including those gifted from another laboratory, details of institutional review board or ethics committee approval must be given, and confirmation of written informed consent must be provided if the line is of human origin.
Example of an ethical statement: "The HCT116 cell line was obtained from XXX. The MLH1+ cell line was provided by XXX, Ltd. The DLD-1 cell line was obtained from Dr. XXX. The DR-GFP and SA-GFP reporter plasmids were obtained from Dr. XXX and the Rad51K133A expression vector was obtained from Dr. XXX."
Research Involving Plants
Experimental research on plants (either cultivated or wild) including collection of plant material, must comply with institutional, national, or international guidelines. We recommend that authors comply with the CBD (https://www.cbd.int/convention/) and the CITES (https://cites.org/eng).
For each submitted manuscript supporting genetic information and origin must be provided. For research manuscripts involving rare and non-model plants (other than, e.g., Arabidopsis thaliana, Nicotiana benthamiana, Oriza sativa, or many other typical model plants), voucher specimens must be deposited in an accessible herbarium or museum. Vouchers may be requested for review by future investigators to verify the identity of the material used in the study (especially if taxonomic rearrangements occur in the future). They should include details of the populations sampled on the site of collection (GPS coordinates), date of collection, and document the part(s) used in the study where appropriate. For rare, threatened or endangered species this can be waived but it is necessary for the author to describe this in the cover letter.
Example of an ethical statement: "Torenia fournieri plants were used in this study. White-flowered Crown White (CrW) and violet-flowered Crown Violet (CrV) cultivars selected from ‘Crown Mix’ (XXX Company, City, Country) were kindly provided by Dr. XXX (XXX Institute, City, Country)."
Clinical Trials Registration
Registration: SciMedicine Journal follows the International Committee of Medical Journal Editors (ICMJE) guidelines which require and recommend registration of clinical trials in a public trials registry at or before the time of first patient enrollment as a condition of consideration for publication.
Purely observational studies do not require registration. A clinical trial not only refers to studies that take place in a hospital or involve pharmaceuticals, but also refer to all studies which involve participant randomization and group classification in the context of the intervention under assessment.
Authors are strongly encouraged to pre-register clinical trials with an international clinical trials register and cite a reference to the registration in the abstract and Methods section. Suitable databases include clinicaltrials.gov, the EU Clinical Trials Register and those listed by the World Health Organisation International Clinical Trials Registry Platform.
Approval to conduct a study from an independent local, regional, or national review body is not equivalent to prospective clinical trial registration. SciMedicine Journal reserves the right to decline any paper without trial registration for further peer-review. However, if the study protocol has been published before the enrolment, the registration can be waived with the correct citation of the published protocol.
CONSORT Statement: SciMedicine Journal requires a completed CONSORT 2010 checklist and flow diagram as a condition of submission when reporting the results of a randomized trial. Templates for these can be found here or on the CONSORT website (http://www.consort-statement.org) which also describes several CONSORT checklist extensions for different designs and types of data beyond two group parallel trials. At minimum, your article should report the content addressed by each item of the checklist.
- Authors are expected to comply with standard reporting guidelines for study designs. Check the EQUATOR Network for reporting instructions and supporting documentation. Documentation for specific studies should be uploaded as supporting information during manuscript submission. Read the Author Guidelines.
Errata and Corrigenda
Changes/additions to accepted articles
All content of published articles is subject to the editorial review process, organized by and under the auspices of the editor. Should the authors wish to add to their article after acceptance, they must submit a request to the editor and the new content will be reviewed.
- If the new material is added to the accepted article, it must be submitted for peer review as a new manuscript, referring back to the original;
- If the new material should replace the original content of the accepted article, the editor may consider the publication of an erratum or a corrigendum.
An erratum refers to a correction of errors introduced to the article by the publisher.
All publisher-introduced changes are highlighted to the author at the proof stage and any errors are ideally identified by the author and corrected by the publisher before final publication.
CorrigendumA corrigendum refers to a change to their article that the author wishes to publish at any time after acceptance. Authors should contact the editor of the journal, who will determine the impact of the change and decide on the appropriate course of action.
Peer Review Policy
This journal uses double-blind peer-review, which means that the reviewers of the paper won’t get to know the identity of the author(s), and the author(s) won’t get to know the identity of the reviewer. The idea is that everyone should get a similar and unbiased review.
Reviewers’ Responsibilities
(http://publicationethics.org/files/u7140/Peer%20review%20guidelines.pdf)
If SMJ’s Editor has invited you to review a manuscript, please consider the following items:
- Reviewing manuscript critically but constructively and preparing detailed comments about the manuscript to help authors improve their research;
- Reviewing multiple versions of a manuscript as necessary;
- Providing all required information within established deadlines;
- Making recommendations to the editor regarding the suitability of the manuscript for publication in the journal;
- Declaring to the editor any potential conflicts of interest with respect to the authors or the content of a manuscript they are asked to review;
- Reporting possible research misconducts;
- Suggesting alternative reviewers in case they cannot review the manuscript for any reasons;
- Treating the manuscript as a confidential document;
- Not making any use of the work described in the manuscript;
- Not communicating directly with authors, if somehow they identify the authors;
- Not identifying themselves to authors;
- Not passing on the assigned manuscript to another reviewer;
- Ensuring that the manuscript is of high quality and original research;
- Informing the editor if he/she finds the assigned manuscript is under consideration in any other publication to his/her knowledge;
- Writing a review report in English only;
- Authoring a commentary for publication related to the reviewed manuscript.
What should be checked while reviewing a manuscript?
- Novelty;
- Originality;
- Scientific reliability;
- A valuable contribution to science;
- Adding new aspects to the existed field of study;
- Ethical aspects;
- Structure of the article submitted and its relevance to authors’ guidelines;
- References provided to substantiate the content;
- Grammar, punctuation, and spelling;
- Scientific misconduct.
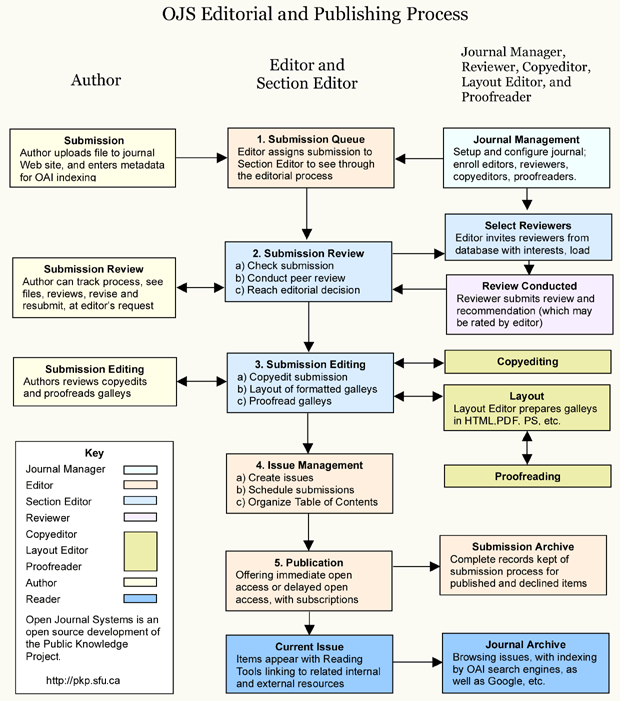
The detailed Journal peer review process is based on the following chart:
Informed Consent Policy
Studies on patients or volunteers require ethics committee approval and informed consent, which should be documented in the paper. Patients have a right to privacy that should not be violated without informed consent. Identifying information, including names, initials, or hospital numbers, should not be published in written descriptions, photographs, or pedigrees unless the information is essential for scientific purposes and the patient (or parent or guardian) gives written informed consent for publication. Informed consent for this purpose requires that an identifiable patient be shown the manuscript to be published.
Authors should disclose to these patients whether any potential identifiable material might be available via the Internet as well as in print after publication. Patient consent should be written and archived with the journal, the authors, or both, as dictated by local regulations or laws. We decide that patient confidentiality is better guarded by having the author archive the consent and instead providing the journal with a written statement that attests that they have received and archived written patient consent. When informed consent has been obtained, it should be indicated in the published article.
Nonessential identifying details should be omitted. Informed consent should be obtained if there is any doubt that anonymity can be maintained. For example, masking the eye region in photographs of patients is inadequate protection of anonymity. If identifying characteristics are de-identified, authors should provide assurance, and editors should so note, that such changes do not distort scientific meaning.
Patient identifiers will not be published in SciMedicine Journal, unless written informed consent is given and the content is essential for the scientific purpose and merit of the manuscript. Photographs of subjects showing any recognizable features must be accompanied by their signed release authorizing publication, as must case reports that provide enough unique identification of a person (other than name) to make recognition possible. Failure to obtain informed consent of patient prior to submission would result in manuscript rejection.
Complaints Policy
We aim to respond to and resolve all complaints quickly and constructively. The procedures to investigate and resolve complaints followed by SciMedicine Journal aims to be fair and balanced for those making complaints and for those being complained about. The complaint can be made by writing an email.
All complaints will be acknowledged within three working days.
Please email to: office@scimedjournal.org










